December 12, 2017
2 min read
New Synthetic DNA Zika Vaccine Effective in Early Trials
By using synthetic DNA to develop new vaccines, researchers can work at a faster time scale than when...
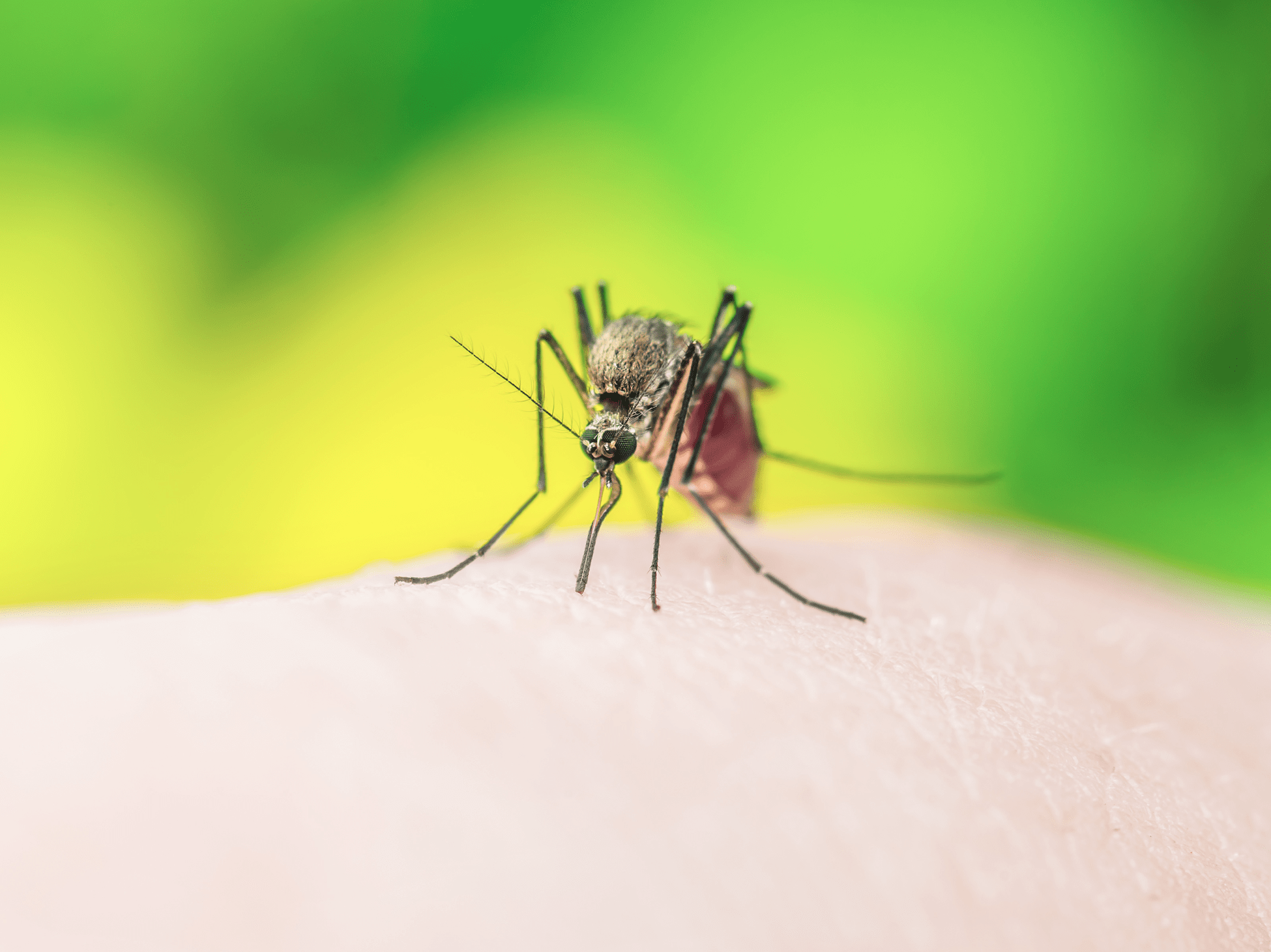
In early October, researchers from the Perelman School of Medicine at the University of Pennsylvania reported that their Zika vaccine candidate successfully induced an immune response among participants in a phase I clinical trial.
Although further research is still required as the vaccine candidate moves through the successive stages of clinical trial, these positive results significantly advance the struggle to develop a vaccine that stops the spread of this epidemic disease.
Unlike traditional vaccines -- which build a recipient’s immunity against a virus by introducing a small amount of viral material and letting the immune system build up antibody defenses -- this new Zika vaccine candidate, GLS-5700, utilizes a novel inoculation method: packaging synthesized portions of the virus. GLS-5700 is a bacterial plasmid that was engineered to contain premembrane and envelope protein from the Zika virus.
In the early trials, these proteins, without any additional viral material, were found to be sufficient to engender an immune response in humans and animal models.
Zika virus was first discovered in Uganda’s Zika forest in 1947. Zika only recently became the subject of international alarm when its spread had reached epidemic proportions in South America, Central America, and the Caribbean islands in 2015 and 2016. Zika is a high-profile epidemic and the subject of widespread panic, even among other mosquito-borne tropical diseases, because of its particularly harmful effects in rare cases. In adults, Zika virus can cause significant neurological effects, such as Guillain-Barré Syndrome. Children born to mothers infected with Zika can be afflicted by microcephaly and other birth defects.
Ever-increasing international travel, especially during such events as the 2016 Brazil Olympics, raised the global urgency of Zika research. Despite several large research efforts, no vaccines have advanced far enough in development to be deemed safe in humans and effective for Zika prevention.
By using synthetic DNA to develop new vaccines, researchers can work at a faster time scale than when they use conventional methods. The accelerated timeframe afforded by synthetic DNA is particularly advantageous in trying to combat emerging infectious diseases and epidemics. With companies like Twist Bioscience providing high-throughput DNA synthesis, the vaccine development timeline can meet the need for disease prevention in high risk scenarios.
The researchers leading the GLS-5700 development believe that synthetic DNA can be utilized in other vaccine development programs.
“Synthetic DNA vaccines, such as this Zika vaccine candidate our team has developed, are an important approach to preventing emerging infectious diseases,” David Weiner, PhD, co-lead author of the synthetic DNA Zika vaccine study, said in a news release. “This novel DNA vaccine was developed and implemented in just months via a platform that has advantages in temperature stability, storage, dose, and distribution compared to most traditional vaccines, making DNA vaccines an important tool to respond quickly to curb an emerging epidemic.”
Chikungunya, West Nile Virus, Pandemic Influenza, and Ebola are targets for similar synthetic DNA vaccines in future research.
What did you think?
Like
Dislike
Love
Surprised
Interesting
Read Next
Get the latest by subscribing to our blog