January 15, 2018
3 min read
Tech Showcase: Anti-CRISPR Protein AcrIIA4
Cas9, when duplexed with a 20-base guide RNA, binds to a sequence matching the reverse transcription of the guide RNA sequence. This property makes Cas9 both precise and powerful.
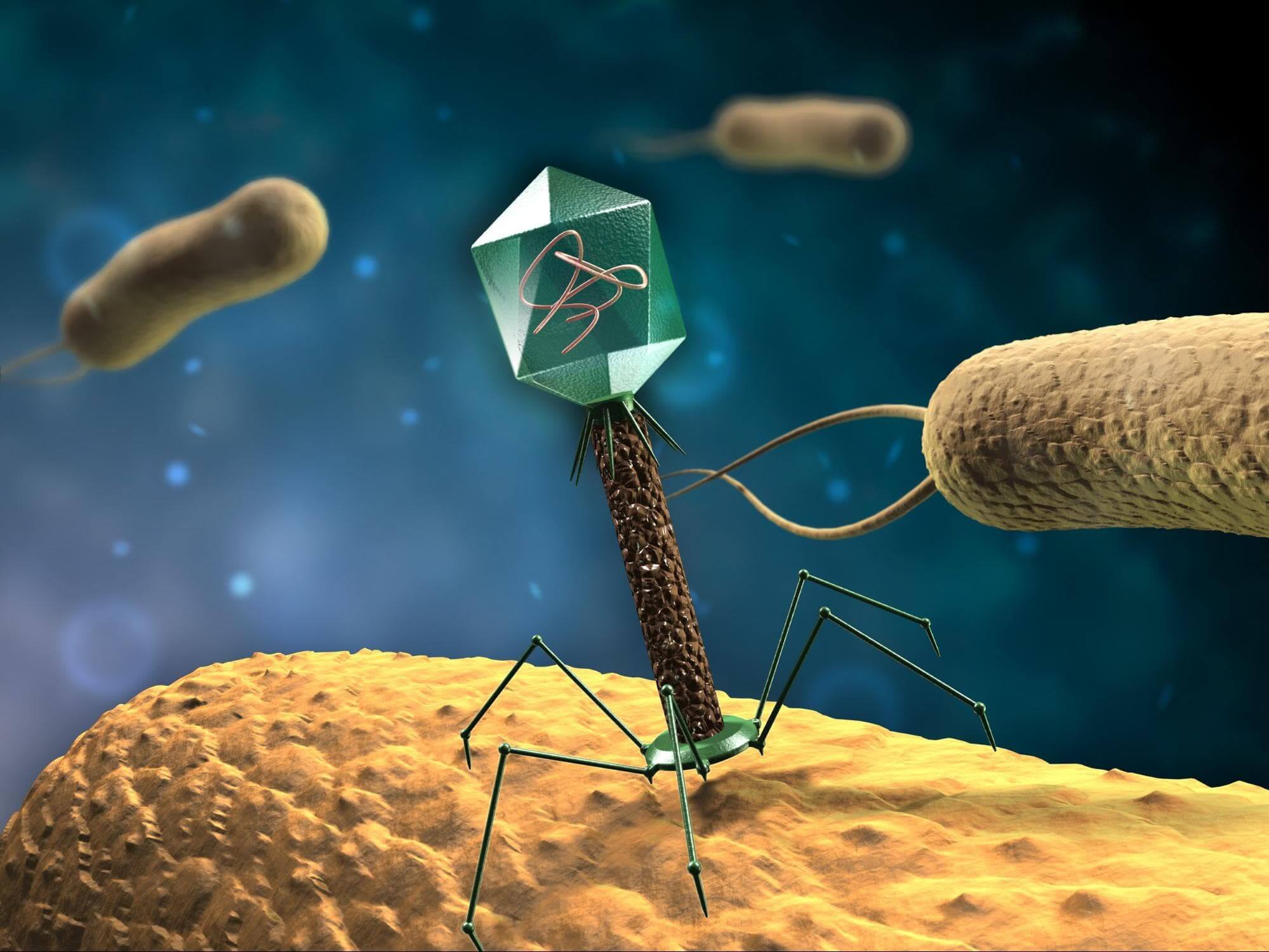
With CRISPR-Cas9’s widespread acclaim as a revolutionizing force in synthetic biology, there are a number of properties that researchers are trying to engineer into the system to gain more complete control over Cas9’s activity.
Cas9, when duplexed with a 20-base guide RNA, binds to a sequence matching the reverse transcription of the guide RNA sequence. This property makes Cas9 both precise and powerful. However, in this Cas9-DNA-RNA system there are numerous concurrent chemical interactions.(1) In 2016, researchers discovered that Cas9 has a lower desired specificity than required for many experiments. This is seen when Cas9 very occasionally binds to sequences that are an imperfect match by one or two bases, resulting in off-target edits.
If this technology is to be used therapeutically in humans like the alternative genome engineering technology TALENs, this mismatching property of Cas9 requires an immediate solution.
On July 12, 2017, a team of researchers led by Jennifer Doudna and Jacob Corn at U.C. Berkeley published a discovery that could add another elegant piece to the expanding set of CRISPR tools. They analyzed the relationship between Cas9 and bacterial immune systems, hoping to elucidate a mechanism by which some bacteria can resist Cas9’s gene-editing.
If you overuse antibiotics, bacteria eventually evolve mechanisms to resist these antibiotics. If bacteria are using an antiviral in the form of CRISPR, it is logical that there are some bacteriophages that have evolved an immunity to Cas9. The researchers searched for viruses able to infect bacteria, despite bacteria possessing complete CRISPR machinery that would typically destroy an invading pathogen.
The researchers found exactly what they went in search for—viruses that were survivors. Then they searched for countermeasures that these viruses deploy to neutralize Cas9. Their hard work paid off—they discovered a small protein called AcrIIA4 that acts as a potent anti-CRISPR molecule. AcrIIA4 is a DNA mimic, which means it inhibits gene alteration by binding strongly to the Cas9-gRNA complex. This blocks the entry of target DNA into the complex, thus preventing Cas9 from reaching its gene target, and inhibiting CRISPR activity.
What sets AcrIIA4 apart and makes it a potential therapeutic asset is that it doesn’t interfere with Cas9’s gene alteration once Cas9 is already bound to the DNA target. In other words, if AcrIIA4 is introduced into cells after Cas9-gRNA has already been delivered and given enough time to reach the target site, then it won’t inhibit ongoing gene alteration, but will halt all alteration thereafter. By controlling the delay between the introduction of Cas9-gRNA and the subsequent introduction of AcrIIA4, the researchers demonstrated that off-target effects could be significantly reduced in human cells, while on-target effects were preserved.
This technology was showcased by study author Joseph Bondy-Denomy at the recent CRISPR conference at Cold Spring Harbour Laboratory. From the talk came a final interesting question: what if bacteria have evolved a secondary mechanism to defend against Cas9 resistant viruses—an anti-anti-CRISPR? Such a molecule would not only allow researchers to have tight control over when to switch off CRISPR, but also switch it on again, providing complete control over the system.
Elucidating AcrIIA4’s mechanism of action and demonstrating its utility in CRISPR editing scenarios broadens CRISPR’s utility. Further examinations of various anti-CRISPR proteins will undoubtedly add more control procedures to the CRISPR toolkit, and contribute to safety protocols that will someday enable therapeutic gene editing. Image: Anti-CRISPR protein AcrIIA4 was discovered in viruses that infect bacteria, like the one depicted above.
(1) There are four interactions between the Cas9 protein and the target DNA, and 20 interactions between the gRNA and target DNA. The free energy within the Cas9-gRNA-DNA system is higher than the free energy required for the system to complete a cut. This leads to an 18-19 bp match between the gRNA and DNA sequences still being energetically permissible for the reaction to continue, albeit at a less efficient rate – the reaction will proceed more slowly when there is less free energy.
What did you think?
Like
Dislike
Love
Surprised
Interesting
Read Next
Get the latest by subscribing to our blog